India has taken another step toward building its own advanced cell-therapy ecosystem, with the Technology Development Board under the Department of Science & Technology signing an agreement with Hyderabad-based Helix Cell Therapeutics Private Limited for the development of a novel dual-targeting CAR-T cell therapy against multiple myeloma. The project, titled “Generation of Novel Dual Targeting Chimeric Antigen Receptor T Cells to Treat Multiple Myeloma and Conducting Phase I Clinical Trial,” will be implemented under an Indo-Singapore collaborative framework with Biocell Innovations, Singapore.
The initiative is significant because multiple myeloma remains a difficult and currently incurable form of blood cancer, especially for patients who relapse after several lines of treatment. Existing CAR-T therapies that target BCMA have already shown encouraging outcomes in relapsed and refractory cases, but the new project aims to go a step further by targeting both BCMA and CD19 markers expressed on multiple myeloma cells.
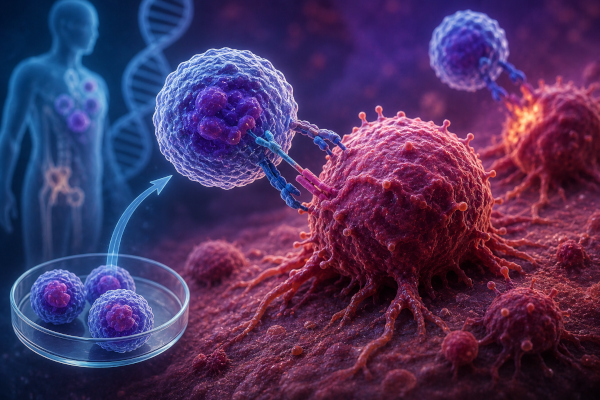
CAR-T cell therapy is one of the most advanced forms of cancer immunotherapy. In this approach, a patient’s own T cells are collected, genetically modified in the laboratory, and trained to recognise specific markers on cancer cells before being infused back into the patient. Once inside the body, these engineered immune cells can identify and attack cancer cells more precisely. Mayo Clinic describes CAR-T as a therapy in which cells from the body are genetically changed so they can fight cancer, especially blood cancers such as leukaemia, lymphoma and multiple myeloma.
Under the TDB-supported project, Helix Cell Therapeutics will develop, manufacture and clinically evaluate the next-generation dual-targeting CAR-T cells through a Phase I clinical trial. The therapy is intended for patients who have exhausted multiple treatment options and have limited therapeutic alternatives available. The dual-targeting strategy is expected to improve the durability of remission compared with conventional single-marker CAR-T approaches.
The focus on BCMA is scientifically important because BCMA is commonly found on plasma cells, the cell type from which multiple myeloma originates, and is often present at higher levels on tumour cells. Research highlighted by the National Cancer Institute has shown that BCMA-targeted CAR-T therapy can produce strong early responses in many patients, although relapse remains a major challenge in the field.
By adding CD19 as a second target, the Indo-Singapore project seeks to reduce the chance of cancer cells escaping treatment through antigen loss or biological variation. This makes the therapy a more ambitious attempt to improve long-term outcomes in difficult-to-treat multiple myeloma patients. The concept is especially relevant at a time when global cancer research is increasingly moving toward more personalised, cell-based and gene-modified therapies.
The project also carries strategic importance for India’s biotechnology sector. It aligns with the Government of India’s push to strengthen indigenous capability in advanced biologics, precision medicine and next-generation healthcare technologies under the broader Aatmanirbhar Bharat framework. It also deepens India-Singapore cooperation in emerging biomedical innovation, an area where international collaboration can speed up clinical development, technology transfer and commercialisation.
TDB Secretary Rajesh Kumar Pathak said advanced cell and gene therapies represent the future of precision healthcare and have transformative potential for treating complex diseases. Helix Cell Therapeutics also said TDB’s support would help accelerate clinical translation and commercialisation of its CAR-T platform while expanding access to advanced cancer-treatment solutions in India.
For Indian patients, the larger promise of such projects lies in affordability and access. Advanced cell therapies are often expensive and technologically complex, limiting their availability in many countries. By supporting domestic development and manufacturing, India can gradually build the scientific, regulatory and industrial base needed to make these treatments more accessible to patients who currently have very few options.
The agreement therefore marks more than a single research grant. It reflects India’s attempt to enter the next frontier of cancer treatment, where engineered immune cells, precision targeting and international collaboration may together open new hope for patients battling some of the toughest blood cancers.
Source: PIB
You may also like
-
Serum Institute of India to Manufacture Oxford Ebola Vaccine in Major Global Health Push
-
India’s Nanomedicine Leap: Pune Scientists Develop Gene-Silencing Platform to Target Breast Cancer Drivers
-
Wockhardt’s Zaynich Opens a New Chapter for Indian Antibiotic Innovation
-
Cinnamon in Ayurveda: The Sweet Heat That Awakens Digestion and Circulation
-
Gingelly / Sesame in Ayurveda: The Tiny Seed That Became Food, Oil and Medicine