In a significant scientific breakthrough, researchers at Hyderabad-based International Advanced Research Centre for Powder Metallurgy and New Materials (ARCI) have resolved a long-standing mystery surrounding the formation of mesoporous tin oxide (SnO₂) beads, a class of advanced materials widely used in gas sensors, lithium-ion batteries and solar cell technologies. The discovery could help scientists and engineers better control particle size, porosity and crystallinity — three factors that strongly influence the performance of these materials in real-world applications.
Mesoporous SnO₂ beads have long attracted attention because of their high surface area and tunable porous structure, making them especially valuable in sensing and energy-storage systems. Until now, however, the exact mechanism by which these beads formed remained unclear. Earlier scientific models had suggested that crystalline nanoparticles emerged during the solvothermal stage itself and later assembled into bead-like structures. The ARCI team has now shown that this explanation was incomplete.
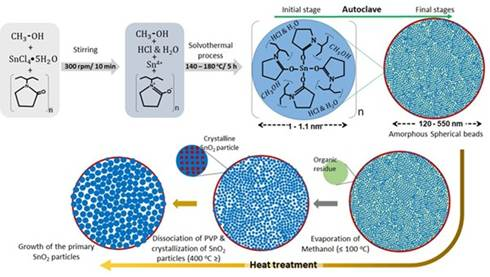
Fig 1: Formation of mesoporous SnO₂ beads from tin-rich complex networks and amorphous spheres formed during stirring and solvothermal treatment, to crystalline primary SnO₂ particles and mesoporous structure development during calcination above 400 °C as PVP decomposes.
According to the findings, the as-prepared beads are not crystalline at all but amorphous in nature. They are made up of a tin-rich organic network containing nanoscale heterogeneities measuring roughly 1.2 to 1.4 nanometres. The study found that primary crystalline SnO₂ particles do not form during the solvothermal process carried out between 140°C and 180°C. Instead, crystallisation begins only during calcination at temperatures of 400°C and above, fundamentally changing the scientific understanding of how these materials evolve.
The researchers further found that, during calcination, polyvinyl pyrrolidone decomposes and generates interconnected voids, which then develop into the characteristic mesoporous architecture of the beads. At the same time, crystallisation and pore formation occur together. The growth process follows the classical Ostwald ripening mechanism, in which larger particles grow at the expense of smaller ones in order to reduce surface energy. A coarsening exponent of around 0.3 confirmed that volumetric diffusion governs this evolution.
A particularly important part of the study was the use of Small Angle X-ray Scattering, or SAXS, which enabled scientists to capture bulk-averaged structural information over sample volumes far larger than what conventional transmission electron microscopy can typically access. This allowed the team to identify nanoscale heterogeneities within the amorphous beads with greater precision and to directly connect microstructural evolution with crystallisation behaviour.
The implications of the work extend well beyond a single material system. By establishing a clearer mechanistic model, the study provides a roadmap for fine-tuning synthesis conditions to produce particles with improved functional properties. That could translate into more sensitive gas sensors, higher-performing lithium-ion batteries and improved performance in advanced solar cell applications. The researchers also noted that the findings position SnO₂ as a useful reference system for understanding the formation of other mesoporous metal oxides such as titanium dioxide, zinc oxide and iron oxide.
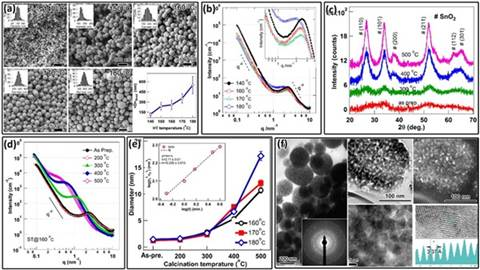
Fig 2 : (a) SEM micrographs showing the morphology of as-prepared beads synthesized at different solvothermal temperatures; (b) SAXS profiles of the as-prepared beads; (c) XRD patterns of beads after heat treatment at various temperatures; (d) SAXS profiles of heat-treated beads highlighting changes in microstructure; (e) Primary particle size as a function of calcination temperature, with the inset depicting Ostwald ripening behavior of beads calcined at 500°C for varying durations; (f) TEM images illustrating the hierarchical structure of beads at different length scales.
The research, published in the Indian Journal of Physics, further strengthens ARCI’s position in advanced materials science and highlights the growing role of Indian institutions in solving fundamental questions with strong technological relevance. Beyond academic interest, the findings may open new opportunities for engineering high-performance materials for energy, environmental and sensing technologies in the years ahead.
Publication link: https://doi.org/10.1007/s12648-024-03419-6)
Source: PIB
You may also like
-
National Institute of Technology Rourkela Develops Patented Bio-Ink for Bone and Cartilage Repair
-
India Identified as Key Centre in Jamun’s Evolutionary History
-
ISRO Targets G20 Climate Satellite in 2027, Space Station by 2035 and Moon Footprints by 2040
-
Indian Study Probes Whether Tiny Dwarf Galaxies May Hide Central Black Holes
-
IIIT Hyderabad Professor Develops AI Chip Architecture Aimed at Lowering Power and Compute Burden
